04 Feb, 2026
Orange, camphor-smelling solid could be a key to the next generation grid-storage batteries
An orange solid with a camphor-like odour has helped aqueous Zinc-Iodide batteries move a large step closer to supplying safe and economic grid and household energy storage.
Researchers from the ARC Centre of Excellence for Carbon Science and Innovation based at Adelaide University have used ferrocene, an electroactive cation and that orange, camphor smelling solid, to solve two key problems plaguing the ability of the aqueous Zinc-Iodide battery to become an alternative energy storage to Lithium-ion batteries: the shuttling effect of reaction intermediates that corrode the anode and the need to boost energy density.
The research is published in Nature Chemistry.
Rechargeable Zinc (Zn) aqueous batteries have attracted attention because of their low cost, affordable energy density and high safety. Porous carbon materials in Zinc-Iodide (Zn-I) batteries, which feature high electronic conductivity and physical adsorption, make them suitable to host iodine to improve the reversibility (charge-discharge) of Zn-I batteries.
The problem is that in Zn-I batteries, the I–/I2 conversion generates soluble polyiodides (essentially a string of iodine atoms) that shuttle to the anode on cycling leading to corrosion of the Zn anode and significant self-discharge.
The porous carbon has typically been used to confine the polyiodides, but this adsorption is weak and shuttling effects cannot be completely avoided. Limiting the amount of iodide in the porous carbon has been used as a tradeoff but that compromises the energy density. Other cations employed to confine the polyiodides have been electrochemically inactive.
Centre of Excellence Deputy Director and Chief Investigator, Prof Shizhang Qiao and his team developed an electroactive redox coupling strategy that introduced ferrocene, an electroactive, organometallic compound, into the I2 cathodes.
“The Ferrocene effectively captures the polyiodides transforming the soluble polyiodide-mediated liquid phase into an insoluble ferrocenium-polyiodide complex that stops the shuttling effect,” says Professor Qiao.
Without ferrocene, it is only the iodine that provides the energy capacity. But the ferrocene-ferrocenium redox improves the active mass ratios of iodine cathodes from the typically less than 70% in traditional porous carbon to90%,which hassignificantly enhanced the energy density and minimized capacity loss of the batteries.
In addition, Zn-I has very long-term cycling stability that is superior to the Li-ion battery, which will further reduce the overall cost of the batteries.
“As we seek greater reliance on batteries for storage of grid energy, the safety concerns and high cost of Li-ion batteries is creating opportunities for alternative technologies. Aqueous batteries are cheaper, stable and therefore safer, are recyclable and have a longer lifespan,” says Prof Qiao.
“The introduced functional redox materials that reconciled shuttle suppression with high energy density, offers a promising route to the development of advanced aqueous Zn–I2 batteries.
We still have a lot of work to do before commercialisation. At the fundamental level, we are working on improving the energy density further and we are exploring the ability to scale up the system from the ampere-hour (Ah) Zn–I2 pouch cells,” he says.
And the discovery of Ferrocene comes with has some intriquing twists and controversy surrounding Nobel prizes and the first research team who discovered Ferrocene discarding the orangy sludge that formed and blocked the pipeline in a experiment set up for a different discovery. Derek Lowe’s blog goes into the detail.
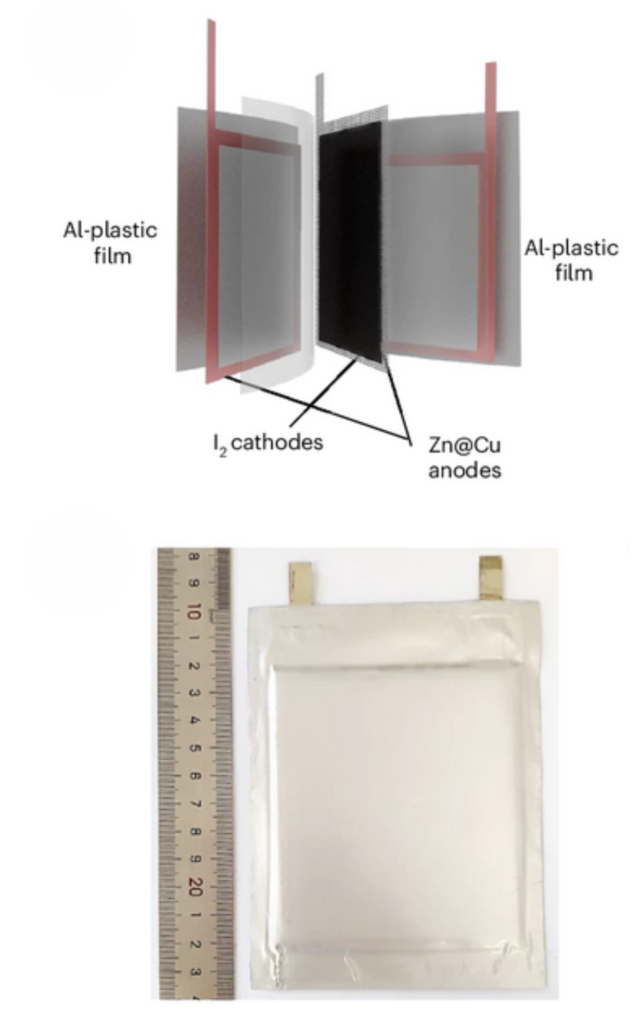
Citation: Zhang, SJ., Hao, J., Wu, H. et al. Electroactive ferrocene/ferrocenium redox coupling for shuttle-free aqueous zinc–iodine pouch cells. Nat. Chem. (2025). https://doi.org/10.1038/s41557-025-01986-7